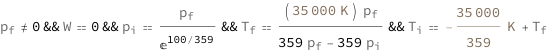Isochoric Process
An isochoric process is a thermodynamic process during which the volume of the closed system undergoing such a process remains constant.
The final temperature divided by the final pressure equals the initial temperature divided by the initial pressure. The initial and final volumes are equal. The work done on the system is zero. The heat transferred to the system equals the isochoric heat capacity times the difference between the final and initial temperatures. The entropy change equals the isochoric heat capacity times the natural logarithm of the ratio of the final temperature to the initial temperature.
Examples
Get the resource:
| Out[1]= |  |
Get the formula:
| Out[2]= | 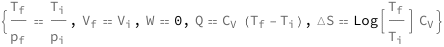 |
Use some values:
| Out[3]= | 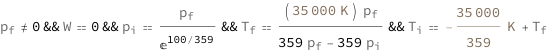 |
External Links
Publisher Information
![Copy to Clipboard {QuantityVariable[Subscript["T", "f"], "Temperature"]/QuantityVariable[Subscript["p", "f"], "Pressure"] == QuantityVariable[Subscript["T", "i"], "Temperature"]/QuantityVariable[Subscript["p", "i"], "Pressure"], QuantityVariable[Subscript["V", "f"], "Volume"] == QuantityVariable[Subscript["V", "i"], "Volume"], QuantityVariable["W", "Work"] == 0, QuantityVariable["Q", "Heat"] == QuantityVariable[Subscript["C", "V"], "HeatCapacity"]*(QuantityVariable[Subscript["T", "f"], "Temperature"] - QuantityVariable[Subscript["T", "i"], "Temperature"]), QuantityVariable["ΔS", "Entropy"] == Log[QuantityVariable[Subscript["T", "f"], "Temperature"]/QuantityVariable[Subscript["T", "i"], "Temperature"]]*QuantityVariable[Subscript["C", "V"], "HeatCapacity"]}](https://www.wolframcloud.com/objects/resourcesystem/marketplacestorage/resources/5e1/5e18aa40-883a-450e-8af9-70c80a111598/Webpage/FormulaImage.png)

![FormulaData[ResourceObject["Isochoric Process"], {QuantityVariable[
\!\(\*SubscriptBox[\("V"\), \("f"\)]\),"Volume"] ->
Quantity[1, ("Meters")^3],
QuantityVariable["Q","Heat"] -> Quantity[70, "Kilojoules"],
QuantityVariable["\[CapitalDelta]\[InvisibleSpace]S","Entropy"] ->
Quantity[200, ("Joules")/("Kelvins")], QuantityVariable[
\!\(\*SubscriptBox[\("V"\), \("i"\)]\),"Volume"] ->
Quantity[1, ("Meters")^3], QuantityVariable[
\!\(\*SubscriptBox[\("C"\), \("V"\)]\),"HeatCapacity"] ->
Quantity[718, ("Joules")/("KelvinsDifference")]}]](images/5e1/5e18aa40-883a-450e-8af9-70c80a111598-io-3-i.en.gif)